Is Enthalpy A Path Function
Land vs. Path Functions
- Folio ID
- 96644
A land role is a belongings whose value does not depend on the path taken to reach that specific value. In dissimilarity, functions that depend on the path from two values are telephone call path functions. Both path and state functions are frequently encountered in thermodynamics.
Introduction
Whenever compounds or chemic reactions are discussed, one of the first things mentioned is the state of the specific molecule or compound. "State" refers to temperature, pressure, and the corporeality and type of substance present. Once the land has been established, state functions can be divers. State functions are values that depend on the state of the substance, and not on how that state was reached. For example, density is a state part, because a substance's density is not affected by how the substance is obtained. Consider a quantity of H2O: it does not thing whether that H2O is obtained from the tap, from a well, or from a canteen, because as long every bit all three are in the same state, they have the same density. When deciding whether a certain belongings is a country function or not, keep this rule in mind: is this property or value affected by the path or manner taken to plant information technology? If the answer is no, and then information technology is a state part, but if the reply is yes, then it is not a state function.
Mathematics of Land Functions
Some other manner to think of state functions is every bit integrals. Integrals depend on only 3 things: the function, the lower limit and the upper limit. Similarly, state functions depend on three things: the property, the initial value, and the final value. In other words, integrals illustrate how state functions depend only on the final and initial value and not on the object'southward history or the path taken to get from the initial to the last value.
Here is an example of the integral of enthalpy, \(H\), where \(t_0\) represents the initial country and \(t_1\) represents the final state.
\[ \displaystyle \int_{t_o}^{t_1} \; H(t) dt = H(t_1)-H(t_o) \]
This is equivalent to a familiar definition of enthalpy:
\[ \Delta H = H_{final} - H_{initial}\]
As represented by the solution to the integral, enthalpy is a land role because information technology simply depends on the initial and final conditions, and not on the path taken to establish these weather. Therefore, the integral of state functions can be taken using only two values: the terminal and initial values. On the other mitt, multiple integrals and multiple limits of integration are required have the integral of a path function. If an integral of a certain holding can be calculated using but the holding and it's initial and final value, the holding is a state function.
State Functions vs. Path Functions
State functions are defined past comparison them to path functions. As stated earlier, a state office is a property whose value does not depend on the path taken to reach that specific function or value. In essence, if something is not a path function, it is probably a land function. To better understand state functions, first define path functions and then compare path and state functions.
Path functions are functions that depend on the path taken to achieve that specific value. For instance, suppose you have $grand in your savings account. Suppose you want to deposit some money to this account. The amount you deposit is a path function because information technology is dependent upon the path taken to obtain that coin. In other words, the amount of money yous volition deposit in your savings account is dependent upon the path or way taken to obtain that money. If y'all piece of work as a CEO of a company for a week versus working at a gas station for a week, you would receive 2 unlike amounts of money at the finish of the week. Thus, a path function is a belongings or value that is dependent on the path taken to plant that value.
State functions do not depend on the path taken. Using the aforementioned instance, suppose you have $1000 in your savings account. Y'all withdraw $500 from your savings business relationship. It does not matter whether y'all withdraw the $500 in i shot or whether you do then at a rate of $50. At the cease when you receive your monthly statement, you will notice a cyberspace withdrawal of $500 and volition encounter your resulting balance as $500. Thus, the banking concern remainder is a state part because it does not depend on the path or way taken to withdraw or eolith money. In the end whether you do so in one lump or in multiple transactions, your bank residual will stay the same. The figure below illustrates state functions in the grade of enthalpy:
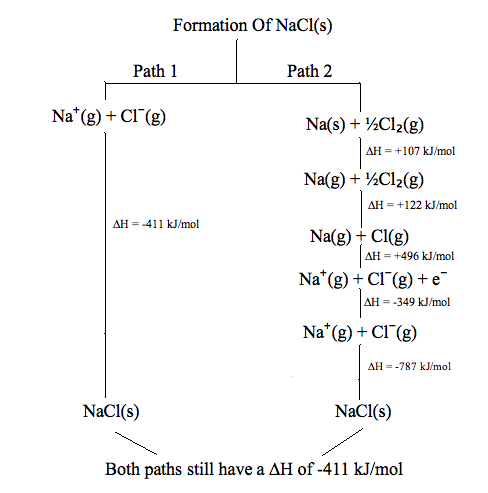
In this figure, 2 dissimilar steps are shown to form \(NaCl_{(due south)}\).
Path i: The commencement path takes merely a single pace with an enthalpy of formation of -411 kJ/mol:
\[Na^+_{(grand)} + Cl^-_{(g)} \rightarrow NaCl_{(s)}\]
Path 2: The second path takes v steps to form \(NaCl_{(s)}\)
\[Na_{(s)} + 1/2 \;Cl_{(g)} \rightarrow Na_{(g)} + 1/2\; Cl_{(yard)} \tag{1: sublimation}\]
\[Na_{(1000)} + one/2 \;Cl_{(thou)} \rightarrow Na_{(thou)} + Cl_{(chiliad)} \tag{2: atomization}\]
\[Na_{(m)} + Cl_{(1000)} \rightarrow Na^+_{(one thousand)} + Cl_{(grand)} \tag{3: ionization}\]
\[Na^+_{(g)} + Cl_{(g)} \rightarrow Na^+_{(g)} + Cl^-_{(g)} \tag{4: electron analogousness}\]
\[Na^+_{(k)} + Cl^-_{(g)} \rightarrow NaCl_{(s)} \tag{5: lattice germination}\]
When enthalpies of all these steps are added, the enthalpy of germination of \(NaCl_{(due south)}\) is still -411 kJ/mol. This is a perfect example of a state function: no matter which path is taken to form \(NaCl_{(due south)}\), it results the same enthalpy of germination of -411 kJ/mol.
| State Office | Path Function |
|---|---|
| Independent of path taken to establish property or value. | Dependent on path taken to establish property or value. |
| Can integrate using final and initial values. | Demand multiple integrals and limits of integration in society to integrate. |
| Multiple steps result in same value. | Multiple steps result in different value. |
| Based on established land of system (temperature, pressure, amount, and identity of system). | Based on how state of system was established. |
| Usually represented past an uppercase letter.one | Commonly represented by a lowercase letter.1 |
1The final comparison fabricated is a generalization that does not necessarily hold for all aspects and calculations involved in chemistry.
Illustration
The chief signal to call up when trying to identify a state function is to make up one's mind whether the path taken to accomplish the function affects the value. The analogy below illustrates how to tell whether a certain property is a state function.
Every morning, millions of people must decide how to reach their offices. Some opt for taking the stairs, whereas others have the elevator. In this situation, ∆y, or change in vertical position is the same whether a person accept the stairs or the elevator. The distance from the function anteroom to the function stays the aforementioned, irrespective of the path taken to get to your office. Every bit a result, ∆y is a country function because its value is independent of the path taken to establish its value.
In the same situation, fourth dimension, or ∆t, is non a state office. If someone takes the longer way of getting to the office (climbing the stairs), ∆t would exist greater, whereas ∆t would be smaller if the elevator is taken. In this illustration, ∆t is not a state function because its value is dependent on the path.
Applications
State functions are commonly encountered in thermodynamics; many of the equations involved with thermodynamics, such as \(\Delta U\) and \(\Delta H\), are state functions. Additionally, state functions are crucial in thermodynamics because they brand calculations simple and permit one to calculate data that could otherwise simply exist obtained through experiments.
More specifically, state functions facilitate the apply of Hess'south Police, which allows the manipulation (improver, subtraction, multiply etc.) of the enthalpies of one-half reactions when adding multiple half reactions to form a full reaction. Hess's Law is dependent upon the fact that enthalpy is a state function. If enthalpy was non a state role, Hess's Police would be much more complicated, because the enthalpies of one-half reactions could not be added. Instead, several additional calculations would be required. Furthermore, state functions and Hess's Police force helps one calculate the enthalpy of circuitous reactions without having to really replicate these reactions in a laboratory. All that is required is to write out and sum the enthalpy of the one-half reactions or of the hypothetical steps leading to the chemical reaction. State functions are too encountered in many other equations involved with thermodynamics such every bit internal energy (∆U), Gibb's costless energy, enthalpy, and entropy.
Issues
- In terms of what we accept discussed in this module, is going from the 1st flooring of Sproul hall, to the 9th floor of Sproul hall, the aforementioned affair as going from the 1st floor of Sproul hall, to the 3rd floor, to the 5th floor, to the 9th floor of Sproul hall?
- Is ∆U a state function?
- Is temperature a land function?
- Is book a state function? (prove with an case)
- Although pressure and book are state functions, why is work (which is ofttimes expressed as -P∆V) not a state role?
Solutions
- Yes, because the question describes a state office. Your position is dependent only on the last and initial position, which are respectively 9th floor of Sproul and 1st flooring of Sproul, and not on the path or mode taken to go there.
- The formula for ∆U is, ∆U = Ulast- Uinitial . The formula of ∆U itself proves that it is a land function considering ∆U is only dependent on Uconcluding and Uinitial . In other words, ∆U is not afflicted by the path taken to establish its values. This is the definition of a land function and equally a result, ∆U is a state office.
- Temperature is a state function as it is one of the values used to define the land of an object. Furthermore, temperature is dependent on the final and initial values, not on the path taken to constitute the values.
- Volume is a country office because book is merely dependent on the final and initial values and not on the path taken to establish those values. Any example that shows this statement in role is adequate. Here's an acceptable respond: Imagine a balloon is inflated until a sure volume. If inflated in multiple steps or in a unmarried stride, it will all the same attain the aforementioned book at the stop. As a result, volume is a state part because it is non dependent on the object's path or history.
- The reason work is non a state part depends on the definition of work rather than the formula of work. The definition of work is moving an object against a strength. Thus, in essence, the definition of work states that work depends on its history or path it takes considering the movement of an object is dependent upon the path taken to execute that motion (i.due east. running vs. walking). Therefore, if an object is dependent on its history or on the path information technology takes, the resulting value or belongings is not a state part. Even though pressure level and book are state functions, the definition of work illustrates why work is non a country function.
References
- Petrucci, Ralph H., Harwood, William Southward., Herring, F. G., and Madura Jeffrey D. General Chemistry: Principles & Modern Applications. 9th Ed. New Bailiwick of jersey: Pearson Education, Inc., 2007. Print.
- Kotz, John C., Treichel, Paul 1000., and Townsend, John. Chemistry & Chemical Reactivity. 7th Ed. Belmont: Thomson Higher Education, 2006. Impress.
Contributors and Attributions
- Allison Billings (UCD), Rachel Morris (UCD), Ryan Starr (UCD), Angad Oberoi (UCD)
Is Enthalpy A Path Function,
Source: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_%28Physical_and_Theoretical_Chemistry%29/Thermodynamics/Fundamentals_of_Thermodynamics/State_vs._Path_Functions
Posted by: scottwomell1958.blogspot.com


0 Response to "Is Enthalpy A Path Function"
Post a Comment